 
This would mean that indium’s atomic mass was actually 113, placing the element between two other metals, cadmium and tin. I hope you have got the complete list of 118 elements and their symbols and atomic number along with their atomic mass. Order generally coincides with increasing atomic mass. Visualize trends, 3D orbitals, isotopes, and mix compounds. Because elemental indium is a silvery-white metal, however, Mendeleev postulated that the stoichiometry of its oxide was really In 2O 3 rather than InO. The elements whose atomic masses are written in bracket ( ) are the synthetic elements and their atomic masses values represent the Atomic Mass of the most stable isotope. Elements are arranged from left to right and top to bottom in order of increasing atomic number. Interactive periodic table showing names, electrons, and oxidation states. If this atomic mass were correct, then indium would have to be placed in the middle of the nonmetals, between arsenic (atomic mass 75) and selenium (atomic mass 78). The atomic mass of indium had originally been reported as 75.6, based on an assumed stoichiometry of InO for its oxide. However, by simply multiplying an atomic mass by 1 g/mol, a workable quantity is obtained for an element's molar mass - the mass (in grams. Atomic masses, when expressed in amu, as on the periodic table, are technically unitless. He discovered, for example, that the atomic masses previously reported for beryllium, indium, and uranium were incorrect. Relative atomic masses, as listed on the periodic table, are used to calculate molar masses for atoms and molecules. Core share and HTML view are not possible as. 1 Orientation: what is physical chemistry about. When the chemical properties of an element suggested that it might have been assigned the wrong place in earlier tables, Mendeleev carefully reexamined its atomic mass. Appendix F - Periodic table of the elements, with molar masses. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. The molar mass is an average of many instances of the compound, which often vary in mass due to. 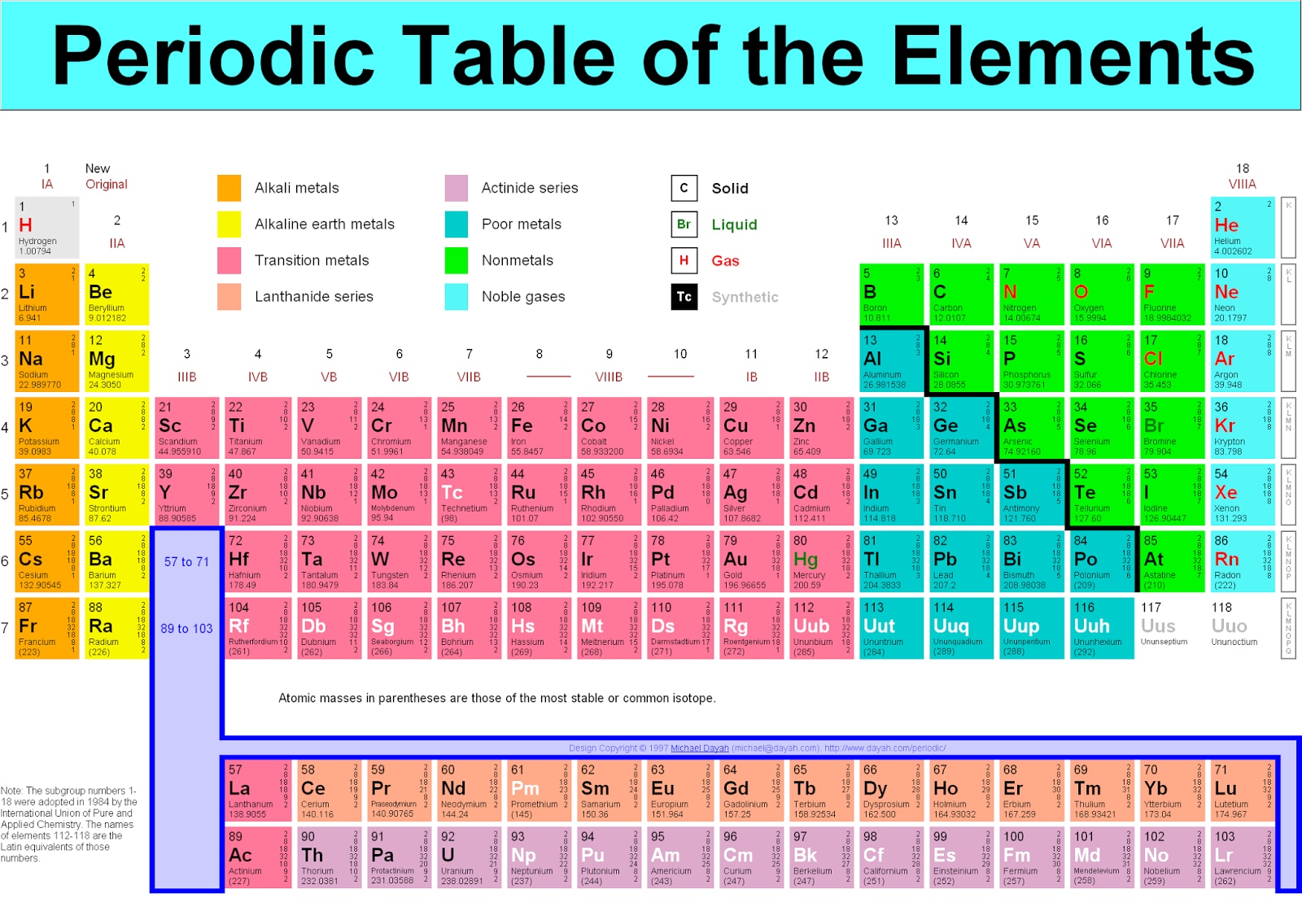
1 The molar mass is a bulk, not molecular, property of a substance. Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole.\): Comparison of the Properties Predicted by Mendeleev in 1869 for eka-Aluminum and eka-Silicon with the Properties of Gallium (discovered in 1875) and Germanium (discovered in 1886). In chemistry, the molar mass ( M) of a chemical compound is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of said compound. The division sign (/) implies “per,” and “1” is implied in the denominator. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). The Molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. adenosine (C 10H 13N 5O 4), a component of cell nuclei crucial for cell divisionīe careful when counting atoms.barium sulfate (BaSO 4), used to take X rays of the gastrointestional tract.What is the mass of 1 mol of each substance? \): Moles to Mass Conversion with Compounds 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
